Difference Between Acetone And Acetic Acid
 Ava Arnold
Ava Arnold Acetone and acetic acid are two related compounds that are often confused due to their similar names. Although they are both organic compounds, they have different chemical structures, properties, and uses. Knowing the difference between acetone and acetic acid can help you make the best choice when selecting a product for a specific purpose.
What is Acetone?
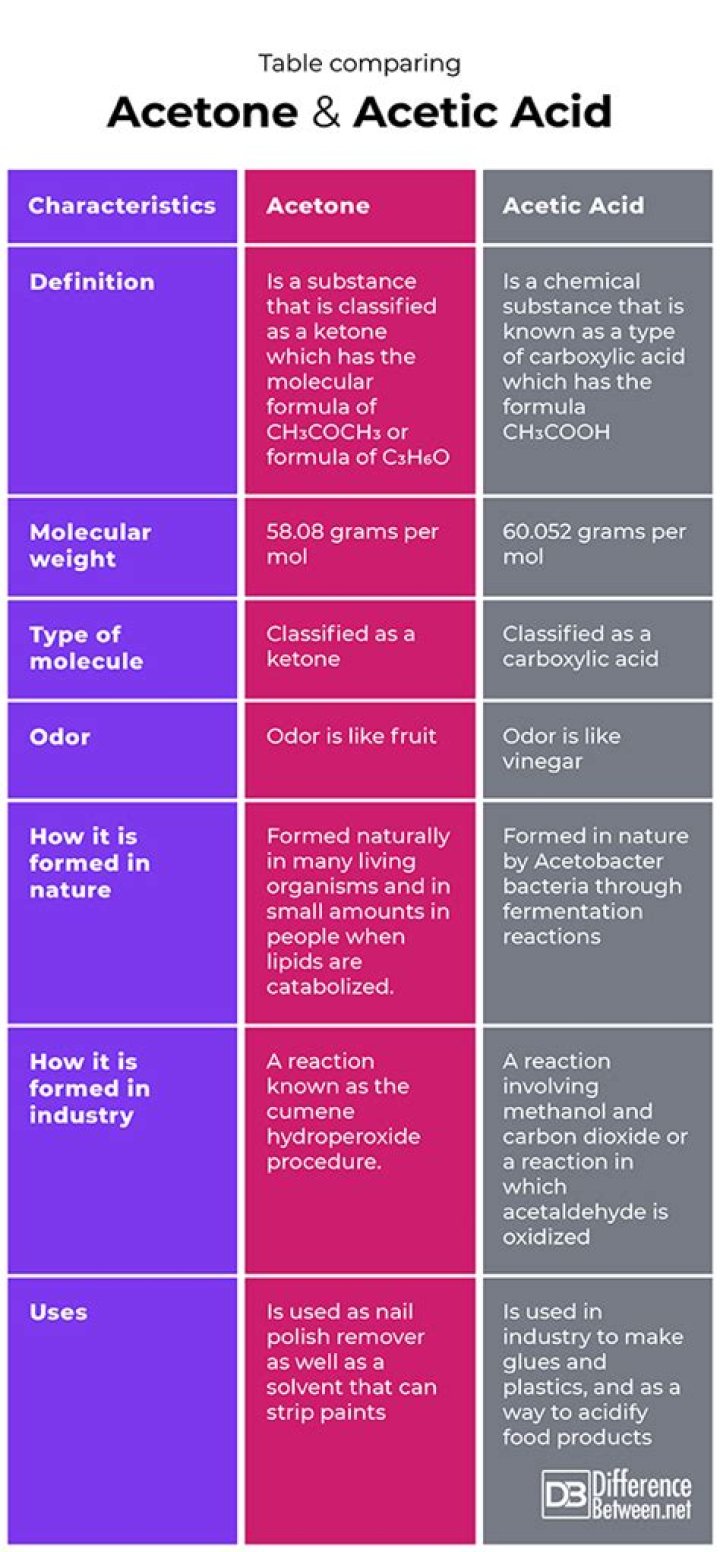
Acetone is an organic compound with the chemical formula C3H6O. It is a colorless, volatile, flammable liquid that evaporates quickly when exposed to air. Acetone is the simplest and smallest ketone, and it is the most commonly used solvent in the world. It is used in a variety of industrial and consumer products, including paint removers, nail polish remover, and adhesives.
What is Acetic Acid?
Acetic acid is an organic compound with the chemical formula C2H4O2. It is a colorless liquid with a strong, pungent odor. Acetic acid is the main component of vinegar and is produced by the natural fermentation of sugars. It is used in a variety of industrial and consumer products, including food preservatives, cleaning products, and pharmaceuticals.
Chemical Structure
The most obvious difference between acetone and acetic acid is their chemical structures. Acetone is a three-carbon ketone, while acetic acid is a two-carbon carboxylic acid. This difference in structure gives the two compounds different properties and uses.
Boiling Point
The boiling point of a compound is an important property that affects its uses. Acetone has a lower boiling point than acetic acid, with a boiling point of 56.2°C compared to 118.1°C for acetic acid. This difference in boiling points makes acetone more volatile and easier to evaporate than acetic acid.
Solubility
The solubility of a compound is another important property that affects its uses. Acetone is more soluble in water than acetic acid, with a solubility of 2.2 g/L compared to 1.5 g/L for acetic acid. This difference in solubility makes acetone more suitable for use in water-based solutions.
Flammability
Acetone is more flammable than acetic acid, with a flash point of −20°C compared to 11°C for acetic acid. This difference in flammability makes acetone more suitable for use in industrial processes where flammable solvents are needed.
Odor
The odor of a compound is an important property that affects its uses. Acetone has a characteristic sweet, pungent odor, while acetic acid has a strong, pungent odor. This difference in odor makes acetone more suitable for use in products where odor is important.
Toxicity
The toxicity of a compound is an important property that affects its uses. Acetone is less toxic than acetic acid, with an oral LD50 of 6,700 mg/kg compared to 1,000 mg/kg for acetic acid. This difference in toxicity makes acetone more suitable for use in products where toxicity is a concern.
Uses
The uses of acetone and acetic acid are also different. Acetone is used as a solvent in a variety of industrial and consumer products, including paint removers, nail polish remover, and adhesives. Acetic acid is used in a variety of industrial and consumer products, including food preservatives, cleaning products, and pharmaceuticals.
Storage
The storage of acetone and acetic acid is also different. Acetone should be stored in a cool, dry place away from heat and direct sunlight. Acetic acid should be stored in a cool, dry place away from heat, direct sunlight, and other sources of ignition.
Safety
The safety of acetone and acetic acid is also different. Acetone is highly flammable and can cause skin irritation. Acetic acid is corrosive and can cause skin and eye irritation. When handling either compound, it is important to wear protective clothing and to use proper safety equipment.
Production
The production of acetone and acetic acid is also different. Acetone is produced by the oxidation of isopropanol, while acetic acid is produced by the fermentation of sugars or by the oxidation of acetaldehyde. This difference in production makes acetone more expensive to produce than acetic acid.
Price
The price of acetone and acetic acid is also different. Acetone is more expensive than acetic acid, with a price of around $3.50 per gallon compared to around $1.50 per gallon for acetic acid. This difference in price makes acetone less cost-effective for many uses.
Conclusion
Acetone and acetic acid are two related compounds that have different chemical structures, properties, and uses. Knowing the difference between acetone and acetic acid can help you make the best choice when selecting a product for a specific purpose. Acetone is more flammable, volatile, and expensive than acetic acid, while acetic acid is more corrosive and toxic.